Updated Phase 2 OraGrowtH Data Presented at ESPE 2024 Demonstrate Sustained Growth on Oral LUM-201 to 24 Months in PGHD and Correlation of Growth to LUM-201’s Unique Pulsatile Mechanism of Action
“The new analyses and updated data from our Phase 2 OraGrowtH210 and OraGrowtH212 Trials presented at ESPE this week demonstrate sustained growth on LUM-201 in moderate Pediatric Growth Hormone Deficiency (PGHD) to 24 months, as well as the correlation of LUM-201’s pulsatile mechanism of action (MOA) to growth,” said
In the oral presentation of Abstract FC7.6 on
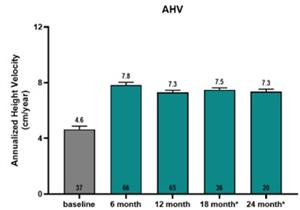
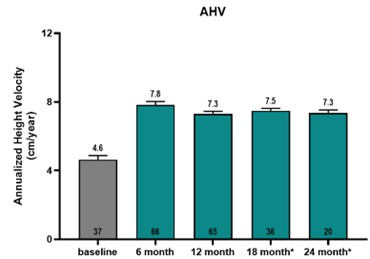
- Results showed that at six months on LUM-201, a significant increase over baseline in Annualized Height Velocity (AHV) was observed for the combined 1.6 and 3.2 mg/kg/day doses. At baseline, AHV was 4.6 cm/yr for 37 subjects for which baseline data were available vs 7.8 cm/yr at 6 months on LUM-201 for 66 subjects.
- Results also showed that growth rates on oral LUM-201 (combined 1.6 and 3.2 mg/kg dose cohorts) were sustained out to 24 months with AHVs of 7.3 (N=65), 7.5 (N=36), and 7.3 (N=20) cm/yr at 12, 18, and 24 months, respectively [graphic below].
- Combined results also demonstrated significant increases in IGF-1 and IGFBP-3 levels from baseline out to 24 months, with IGF-1 increasing from 13.1 (baseline) to 25.5 (12 months) to 29.2 (24 months) nmol/L and IGFBP-3 increasing from 133.5 (baseline) to 179.6 (12 months) to 186.2 (24 months) nmol/L.
- Conclusions: LUM-201 shows a significant increase in AHV at 6 months, and the effect continues with a minimal decrease in AHV through 24 months. Compared to historical rhGH (KIGS)1, LUM-201 has a smaller drop in AHV from 12 months to 24 months. LUM-201 significantly increases IGF-1 and IGFBP-3 levels at 12 months which continues to 24 months. A favorable investigational safety profile has been observed to date.
In the late-breaking oral presentation of Abstract FC15.2 on
- Data from 22 prepubertal children with moderate PGHD from the OraGrowtH212 Trial were evaluated with subjects grouped into tertiles based on 6-month annualized height velocity (AHV).
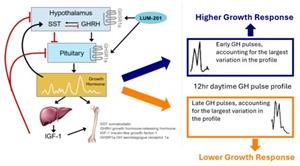
- Pulsatile growth hormone (GH) pulse assessments at baseline and at 6 months (M6) were characterized using 1) Approximate Entropy (ApEn, scale 0-1) examining degree of orderliness over the whole profile and 2) Functional Principal Component Analysis (FPCA) identifying where within the 12-hour profile, divided into 4-hour periods, the dominant variation occurred.
- Results of 6-month treatment with LUM-201:
- Pulsatile GH secretion, ApEn, IGF-1 SDS and IGFBP-3 SDS increased from baseline to M6.
- Greater M6 AHV was associated with an earlier periodicity of augmented pulsatile GH secretion [graphic below].
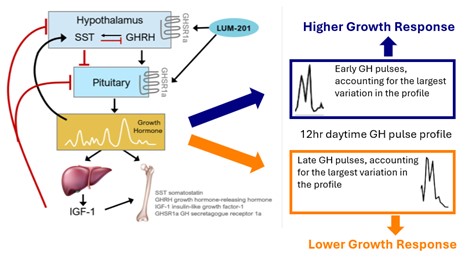
- Conclusion: Both amount and pattern of GH secretion are important for growth and IGF-1 responses to LUM-201.
1 Ranke et al., 2010 – Pfizer KIGS database rhGH treated cohort of moderate prepubertal GHD children.
About
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements of Lumos Pharma, Inc. that involve substantial risks and uncertainties. All such statements contained in this press release are forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. A law that, in part, gives us the opportunity to share our outlook for the future without fear of litigation if it turns out our predictions were not correct.
We are passionate about our business - including LUM-201 and the potential it may have to help patients in the clinic. This passion feeds our optimism that our efforts will be successful and bring about meaningful change for patients. Please keep in mind that actual results or events could differ materially from the plans, intentions and expectations disclosed in the forward-looking statements that we make.
We have attempted to identify forward-looking statements by using words such as “projected,” “upcoming,” “will," “would,” “plan,” “intend,” “anticipate,” “approximate,” “expect,” “potential,” “imminent,” and similar references to future periods or the negative of these terms. Not all forward-looking statements contain these identifying words. Examples of forward-looking statements include, among others, statements we make regarding the advancement of oral LUM-201 to Phase 3, the potential for LUM-201 to be the first oral therapeutic for PGHD, and any other statements other than statements of historical fact.
We wish we were able to predict the future with 100% accuracy, but that just is not possible. Our forward-looking statements are neither historical facts nor assurances of future performance. You should not rely on any of these forward-looking statements and, to help you make your own risk determinations, we have provided an extensive discussion of risks that could cause actual results to differ materially from our forward-looking statements including risks related to the timing and ability of
We anticipate that subsequent events and developments will cause our views to change. We may choose to update these forward-looking statements at some point in the future, however, we disclaim any obligation to do so. As a result, you should not rely on these forward-looking statements as representing our views as of any date subsequent to the date of this press release.
Investor & Media Contact:
Lumos Pharma Investor Relations
512-792-5454
ir@lumos-pharma.com
Photos accompanying this announcement are available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/f1abdd2c-307b-47bb-8cc9-3c555fe00c85
https://www.globenewswire.com/NewsRoom/AttachmentNg/b168603b-a009-4bce-a4d1-94367f1568ec
Source:

Source: Lumos Pharma, Inc.